How the I-neb AAD System Works |

The I-neb® AAD® System uses Adaptive Aerosol Delivery (AAD) technology to precisely and reproducibly deliver VENTAVIS® (iloprost) Inhalation Solution to every patient, at either dose (2.5 mcg or 5.0 mcg), using a single ampule of medication. The I-neb AAD System monitors a patient's unique breathing pattern and adapts to breathing changes during treatment. The I-neb AAD System delivers VENTAVIS in the form of a mist only upon inhalation.2
With the I-neb AAD System, a medication chamber controls the dose and a control disc operates the I-neb AAD System. Once a patient receives a complete dose, the I-neb AAD System stops delivering aerosol and signals the user with visual and audible indicators.
Maximum lung deposition2,16
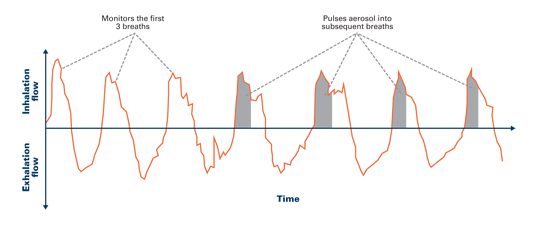
I-neb AAD analyzes breathing patterns for precision dosing
- Determines starting point for aerosol delivery by measuring pressure changes in airflow2
- Sets duration of each pulse by analyzing breathing pattern2
- Continually adapts aerosol delivery to changes in breathing patterns2,16
- Less aerosol waste with I-neb AAD as compared with conventional nebulizers2
Adapted from Nikander16
Based on an in vitro study.
I-neb generates micro-aerosol and analyzes breathing patterns, delivering the drug during only the first 80% of inhalation to maximize lung deposition.2,16
INDICATION
VENTAVIS® (iloprost) Inhalation Solution is indicated for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1) to improve a composite endpoint consisting of exercise tolerance, symptoms (NYHA Class), and lack of deterioration. Studies establishing effectiveness included predominantly patients with NYHA Functional Class III-IV symptoms and etiologies of idiopathic or heritable PAH (65%) or PAH associated with connective tissue disease (23%).
IMPORTANT SAFETY INFORMATION
WARNINGS AND PRECAUTIONS
Risk of Syncope
- Vital signs should be monitored while initiating VENTAVIS. Hypotension leading to syncope has been observed; VENTAVIS should therefore not be initiated in patients with systolic blood pressure less than 85 mmHg.
Pulmonary Venous Hypertension
- Stop VENTAVIS immediately if signs of pulmonary edema occur; this may be a sign of pulmonary venous hypertension.
Bronchospasm
- VENTAVIS inhalation may cause bronchospasm and patients with a history of hyperreactive airway disease may be more sensitive.
ADVERSE REACTIONS
Serious Adverse Events
- Serious adverse events reported include congestive heart failure, chest pain, supraventricular tachycardia, dyspnea, peripheral edema, and kidney failure.
Adverse Events
- Adverse events reported in a Phase 3 clinical trial occurring with a ≥3% difference between VENTAVIS patients and placebo patients were vasodilation (flushing) (27% vs 9%), increased cough (39% vs 26%), headache (30% vs 20%), trismus (12% vs 3%), insomnia (8% vs 2%), nausea (13% vs 8%), hypotension (11% vs 6%), vomiting (7% vs 2%), alkaline phosphatase increased (6% vs 1%), flu syndrome (14% vs 10%), back pain (7% vs 3%), tongue pain (4% vs 0%), palpitations (7% vs 4%), syncope (8% vs 5%), GGT increased (6% vs 3%), muscle cramps (6% vs 3%), hemoptysis (5% vs 2%), and pneumonia (4% vs 1%).
DRUG INTERACTIONS
Antihypertensives and Vasodilators
- VENTAVIS has the potential to increase the hypotensive effect of vasodilators and antihypertensive agents.
Anticoagulants and Platelet Inhibitors
- VENTAVIS also has the potential to increase risk of bleeding, particularly in patients maintained on anticoagulants or platelet inhibitors.
SPECIFIC POPULATIONS
Lactation
- Advise not to breastfeed during treatment with VENTAVIS.
Please see full Prescribing Information.
cp-134777v3

